What Is Diphenhydramine Hydrochloride Powder?
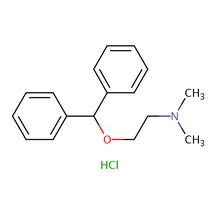
Key Properties
| Molecular Formula | C₁₇H₂₁NO · HCl |
| Molecular Weight | 291.82 g/mol |
| CAS Number | 147-24-0 (Hydrochloride Salt); 58-73-1 (Free Base) |
| Appearance | A fine, white to off-white, crystalline powder. It is odorless or has a faint, characteristic odor. |
| Solubility | Freely soluble in water, ethanol, and chloroform. Slightly soluble in acetone. Practically insoluble in ether and benzene. This high water solubility is crucial for its formulation into various dosage forms like syrups, injections, and capsules. |
Product Specifications
|
Parameter |
Specification / Requirement |
|
Assay (Purity) |
98.0% - 102.0% |
|
Identification |
Conforms (by IR spectroscopy, TLC, or chemical reaction) |
|
Loss on Drying |
≤ 0.5% |
|
Residue on Ignition (Sulfated Ash) |
≤ 0.1% |
|
Heavy Metals |
≤ 20 ppm |
|
Related Substances |
Individual impurities and total impurities within specified limits (controlled by HPLC) |
|
Microbial Limits |
Complies with tests for absence of E. coli, Salmonella, and specified total microbial counts. |
|
Particle Size |
Can be customized (e.g., micronized) for specific formulation needs. |
|
Shelf Life |
Typically 36 months from date of manufacture when stored in original, unopened containers. |
|
Storage |
Preserve in well-closed, light-resistant containers at a controlled room temperature (15-30°C). |
How It Works
Diphenhydramine Hydrochloride Powder is a histamine H1-receptor antagonist, meaning it blocks the action of histamine, a chemical released in allergic reactions. By blocking H1 receptors, it helps reduce symptoms caused by histamine release such as itching, swelling, and sneezing.
It also has anticholinergic (drying) and sedative effects, which is why many allergy medications with diphenhydramine can cause drowsiness, and why it's sometimes used as a short-term sleep aid.
Common Uses of Diphenhydramine Hydrochloride Powder
Allergy Relief
Used in formulations for treating seasonal allergies, hay fever, hives, and itching.
Cold & Flu Support
Included in multi-symptom cold and flu products to reduce runny nose, sneezing, and other symptoms.
Sleep Aid Formulations
Its sedative action makes it common in OTC sleep-promoting products.
Motion Sickness & Nausea
Effective in reducing nausea and vomiting associated with motion sickness when included in appropriate formulations.
Pharmaceutical Ingredient
The powder is used as an active pharmaceutical ingredient (API) in manufacturing tablets, capsules, syrups, and compounded medicines.
Formulation Applications & Compatibility
This versatile API is formulated into numerous dosage forms, each with specific excipient considerations:
Oral Solids (Tablets, Capsules):
Common Excipients: Lactose, microcrystalline cellulose, starch, povidone (binder), magnesium stearate (lubricant), croscarmellose sodium (disintegrant).
Note: Compatibility with reducing sugars (like lactose) should be checked under stress conditions to avoid potential Maillard reaction (browning).
Oral Liquids (Syrups, Elixirs):
Common Excipients: Simple syrup, glycerin, sorbitol, ethanol, flavorings, preservatives (e.g., sodium benzoate), citric acid for pH adjustment.
Parenteral (Injection):
Must be sterile, pyrogen-free. Formulated in Water for Injection, with potential addition of pH adjusters and stabilizers. The solution must be protected from light.
Topical (Creams, Gels, Sprays):
Used for itch relief. Incorporated into oil-in-water or water-in-oil emulsions with emulsifiers, thickeners, and preservatives.
General Incompatibilities: Strong oxidizing agents, strong bases. In solution, it is incompatible with many alkaloidal salts and may precipitate.
Diphenhydramine Hydrochloride Powder remains a cornerstone API in global pharmaceuticals due to its efficacy, well-understood profile, and versatility. Its manufacture and use embody the intersection of classic organic synthesis, modern pharmaceutical analysis, stringent regulatory science, and evolving public health considerations.
Where to Buy Diphenhydramine Hydrochloride Powder?
TNJONE Pharmaceutical has been deeply involved in the pharmaceutical raw materials field for over ten years and is a high-tech enterprise integrating scientific research, production, and sales. We owns a GMP-compliant factory and specializes in custom-made peptide and intermediate products. Our pharmaceutical ingredients (APIs) that meet CP, USP, BP, and EP pharmacopoeia standards. All of products support third-party testing and certification services. So, when you are looking to purchase Diphenhydramine Hydrochloride Powder, please consider TNJONE Pharmaceutical. We guarantee that our products are of reliable quality and come with relevant certificates of analysis.
Factory Environment
TNJONE factory is located in Huyi District, Shaanxi Province. We strictly adhere to the GMP quality system and have professional QA and QC personnel. We are equipped with advanced analytical, cutting, concentration, purification, freeze-drying, and synthesis equipment. We provide OEM/ODM customized services to meet the diverse product needs of our clients.



TNJONE Products are designed for research and industrial use only. Not intended for clinical or medical purposes.
Hot Tags: diphenhydramine hydrochloride powder cas 147-24-0, China diphenhydramine hydrochloride powder cas 147-24-0 manufacturers, suppliers, factory, Diclofenac Sodium Powder CAS 15307 79 6, Diphenhydramine Hydrochloride Powder, Empagliflozin Powder, Heparin sodium Powder, Heparin Sodium Powder CAS 9041 08 1, L carnitine Powder CAS 541 15 1








